By Kim Smiley
The Patient Safety Network presented a case study where a patient with suspected kidney cancer had the wrong kidney removed. Instead of the right kidney that showed suspected renal cell carcinoma in a CT scan, the healthy left kidney was removed. A second surgery was then performed to remove the right kidney and the patient was left dependent on dialysis after losing both kidneys. The patient wasn’t a candidate for a kidney transplant because of the cancer.
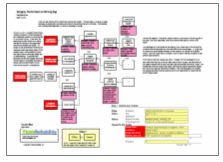
 Reviewing and understanding case studies such as this one is important because wrong-site surgeries are one of the more common serious medical errors. A Cause Map, a visual root cause analysis, can be used to better understand the many causes that contributed to this wrong-site surgery, and better understanding the causes of an incident leads to development of better solutions. The first step in building a Cause Map is to fill in an Outline with the basic background information. These details are often not published for medical errors to protect patient privacy, but the information should be recorded if available. The bottom of the Outline also includes space to list how the issue impacts the overall organizational goals. The Cause Map itself is built by starting at one of the impacted goals and asking “why” questions.
Reviewing and understanding case studies such as this one is important because wrong-site surgeries are one of the more common serious medical errors. A Cause Map, a visual root cause analysis, can be used to better understand the many causes that contributed to this wrong-site surgery, and better understanding the causes of an incident leads to development of better solutions. The first step in building a Cause Map is to fill in an Outline with the basic background information. These details are often not published for medical errors to protect patient privacy, but the information should be recorded if available. The bottom of the Outline also includes space to list how the issue impacts the overall organizational goals. The Cause Map itself is built by starting at one of the impacted goals and asking “why” questions.
Focusing on the patient safety goal as a starting point, the investigation could be started by asking “why was a healthy left kidney removed instead of the right?” The surgeon who performed the surgery believed the tumor was in the left kidney because all patient information readily available stated the tumor was in the left kidney. The case study didn’t include details on how this error in the patient’s record occurred, but it is known that a CT scan was initially performed at a different hospital than the one that performed the surgery. The patient sought treatment at the first hospital after suffering from abdominal pain and hematuria and a CT scan was performed. He was transferred to a second hospital for the surgery after the CT scan revealed suspected renal cell carcinoma. An image of the CT scan was not included with the patient records at the time of transfer and the records noted that there was a tumor in the incorrect (left) kidney.
The stage was essentially set for a wrong-site surgery and the surgeon missed the opportunity to prevent it. The surgeon chose to perform the surgery based on the records without either verifying the original CT (because it was not available) or requesting an additional CT scan to be performed to confirm the diagnosis. It does not appear that the surgeon was required to review the CT scan, but the decision on whether to do so was left up to the surgeon’s judgement. The error was only identified after the pathologist who examined the left kidney found no evidence of cancer and informed the surgeon who then reviewed the original CT scan and realized the wrong kidney had been removed.
Once the causes that contributed to an issue have been identified, the final step in the Cause Mapping process is to identify and implement solutions to prevent a problem from reoccurring. One way to prevent similar errors is to require labeled radiology images to be available to the surgeon prior to any surgery. Requiring a review of images prior to the surgery would build in a double check to ensure the surgery is performed at the correct site. Building in a double check of medical records may also reduce errors like the wrong kidney being listed as potentially cancerous or a patient being transferred with medical files missing important radiology images.