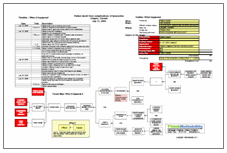
The goal of any root cause analysis is to uncover causes and, most importantly, solutions that will reduce the risk or mitigate the effects of the problem being studied. However, sometimes a problem seems insurmountable. Take rising health care costs. There are myriad causes that contribute to increasing health care costs. Many of the solutions that have been identified are costly, difficult, or both. Additionally, some solutions place the onus on patients, which can limit the effectiveness. Although patients presumably would love to reduce their health care costs, most don’t have the resources to do so.
 Although rising healthcare costs is a national issue, some of the problems you face at an organization may seem just as insurmountable. What can be done when an issue appears too big to fix?
Although rising healthcare costs is a national issue, some of the problems you face at an organization may seem just as insurmountable. What can be done when an issue appears too big to fix?
First, ensure that you limit your analysis and potential solutions to your own sphere of influence. Although patients individually reducing obesity and taking their medications properly and on time would certainly reduce healthcare risks, those steps must be taken by the individuals. As a healthcare organization though, it is possible to take steps to increase the probability that individuals will take these steps. Generally patient education, automated reminders, and making it easier to do the right thing – by including clearer instructions on prescriptions or offering more fresh fruit in the hospital cafeteria – are steps that can be taken that are within the realm of an organization’s sphere of influence. Attempting to control solutions outside your sphere of influence is an exercise in frustration!
Next, focus on a single piece of the pie. Not all the causes identified during a root cause analysis have to be tackled at once. A great way to get started: find a solution that is nearly free and can be implemented fairly transparently to staff. For example, ask providers to hand out a healthy eating brochure to patients as they leave their appointment. Is this going to make a big impact? Probably not, but it’s somewhere to start. And even a little impact can help.
Or, take a note from Camden, New Jersey. In Camden, 1% of patients are responsible for 30% of medical costs. Jeffrey Brenner, a local physician, is making a difference reducing costs by focusing on those few patients. This is the big “bang for your buck” solution. And, the solutions that work for this 1% will probably help reduce costs for the other 99% as well. By focusing on a small number of patients – determining the causes specific to them and tailor-making solutions – headway is being made against an extremely difficult problem. (Read more at: http://www.newyorker.com/online/blogs/newsdesk/2011/01/atul-gawande-super-utilizers.html)
If you’re feeling overwhelmed, try taking one step at a time. If healthcare costs can be tackled by looking at a small part of the problem, what can your organization do with a focused look at solutions?